Last Revised: May 15, 2023
Original version 12/2018, Revised 1/2019, 3/2019, 3/2021
Note: For the purpose of this Fact Sheet, the term "disposable nitrile gloves" refers to gloves that are one-time use and have a thickness of 5-mil and less. Examples are given in the tables at the end of the fact sheet.
Nitrile is a synthetic rubber material that offers chemical and abrasion resistance and is a very good general-duty material for gloves.

Thin, disposable, nitrile gloves are the most common gloves used when handling chemicals in laboratories; however, it is important to remember that thin nitrile rubber provides only limited chemical protection. These gloves are intended to be used only as a physical barrier against brief contact with chemicals, and they need to be removed and discarded immediately after they become contaminated.
Glove materials are evaluated on three criteria when exposed to chemicals:
- Breakthrough time: This is how long it takes to detect a substance inside the glove when the outside is exposed to a chemical. A glove that holds ups for greater than eight hours is considered excellent.
- Degradation: This refers to the physical changes in the material such as swelling, cracking, softening or shrinking, which occur when it comes in contact with a chemical. A glove can exhibit chemical breakthrough even if it doesn’t show signs of degradation.
- Permeation rate: This is the rate at which a substance passes through a glove material once breakthrough takes place. This rate includes absorption on the surface, diffusion through the material, and desorption on the inside surface.
It is important to remember that laboratory testing data does not take into account the effects of hand movement and body temperature. The actual protection will be less for a glove that is being worn and stressed during use.
Optimizing Glove Protection
Selecting the right thickness
Glove thickness is usually given in the unit mils, which is equal to one one-thousandth of an inch; therefore, a glove that is 10 mil, is 0.010 inches thick. A thicker gauge glove will provide more protection than a thinner glove of the same material, but often at the expense of touch-sensitivity and dexterity.
Double-gloving can be used to increase the total breakthrough time of a particular glove, but this increases hand fatigue and overheating and is recommended only for short-duration tasks.
Selecting the right size
A glove that is too small for your hand will be over-stretched, which stresses the material and makes it less protective. A glove that is too large may compromise dexterity and may also become more easily damaged during use. Use the manufacturer's size chart to choose an appropriate fit.
Removing gloves carefully
Glove removal (or "doffing") must be done carefully to avoid contact with the outside of the glove. Always wash your hands after disposing of the gloves.
Avoiding cross-contamination
Soiled gloves can contaminate objects and surfaces, later exposing you to chemical hazards. When this occurs, you can unknowingly be exposed to chemical hazards. If you do experience an acute health effect, it can be difficult to determine which chemical was the source of the exposure.
Reduce the likelihood of cross contamination by:
- changing gloves immediately when contaminated and after each chemical-handling task
- discarding gloves immediately after use. Never reuse disposable nitrile gloves!
- removing gloves and washing hands before leaving the lab
- establishing designated glove-only vs. no-glove items such as pens, keyboards, instruments, drawers, door handles, refrigerators, and work spaces
Do not wear gloves in hallways, offices, break rooms, elevators, restrooms, or any other public areas!
A Note about Allergies
In addition to their superior chemical resistance profile, another reason that nitrile gloves are used instead of latex in Penn research labs is to avoid the skin allergies that can be caused by the proteins in the latex rubber. It is possible, however, for some individuals to be sensitive to the accelerators (chemical additives) in nitrile.
Accelerator-free nitrile glove products are available for people who have sensitivity to standard nitrile gloves. One example is pictured here: Hourglass International™ HandPRO™ FreeStyle1100 Nitrile Exam Gloves
This fact sheet provides some general guidelines about the compatibility of nitrile rubber with common lab chemicals. You can print a PDF of the compatibility information to post in your lab. Posting this information is not a requirement. The attachment can be downloaded from the link at the bottom of this Fact Sheet.
In general, nitrile rubber provides short-term splash protection against the following chemicals. Breakthrough will not occur in under 15 mins for a 5-mil or greater thickness glove.
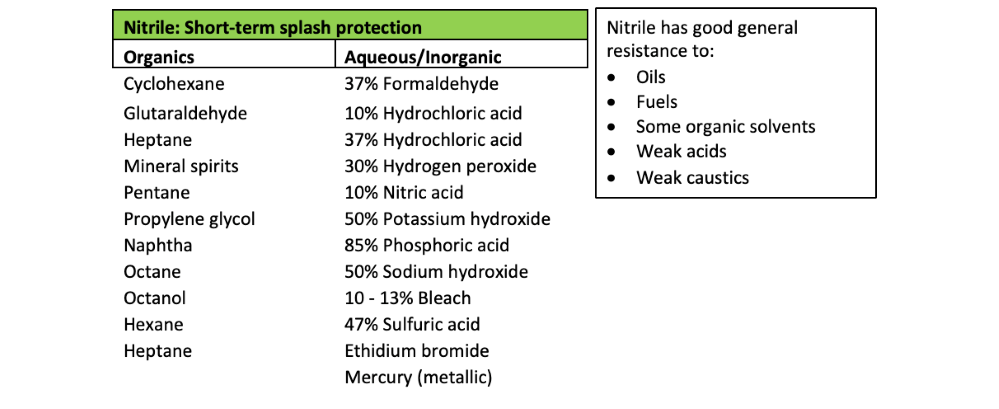
In general, nitrile provides very little protection* against the following common organic chemicals. If gloves come in contact with these chemicals, they must be changed immediately to avoid exposure.
*Breakthrough may occur in under one minute for a 5-mil or lesser thickness glove.


*Disposable nitrile gloves of 8-mil thickness or greater will provide short term (15 minute) splash protection against HF. An appropriately rated chemical-resistant glove is required if prolonged contact with HF is anticipated. For more information about HF see the FACT SHEET: Hydrofluoric Acid in the Chemical Hygiene Plan.
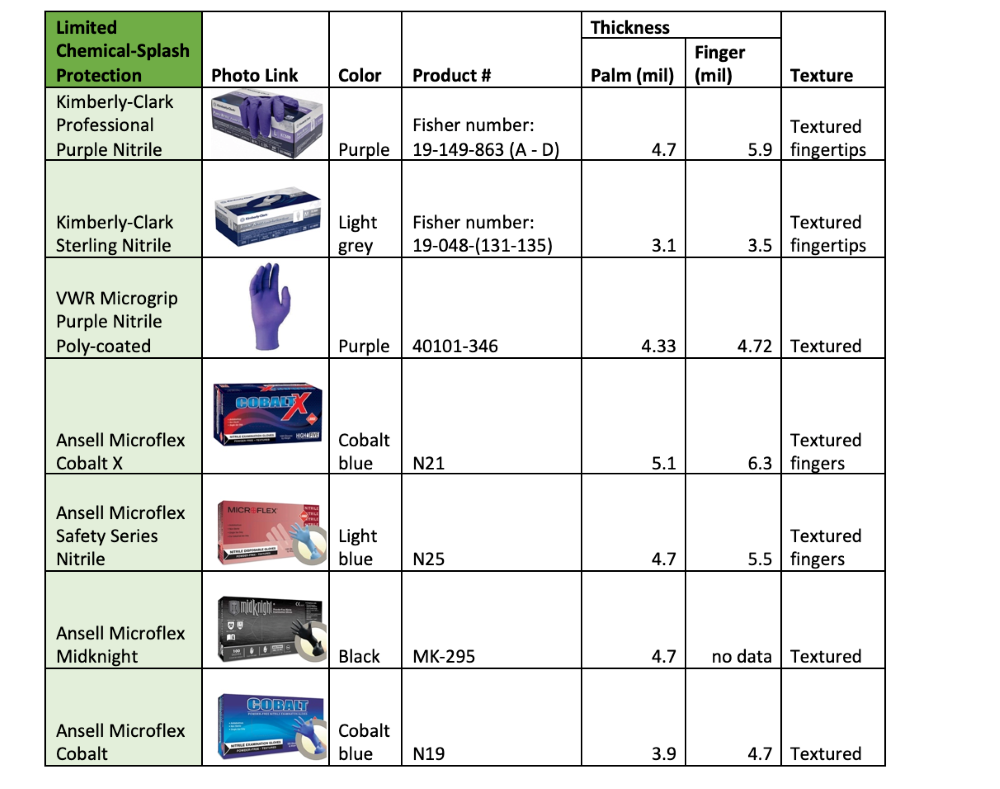
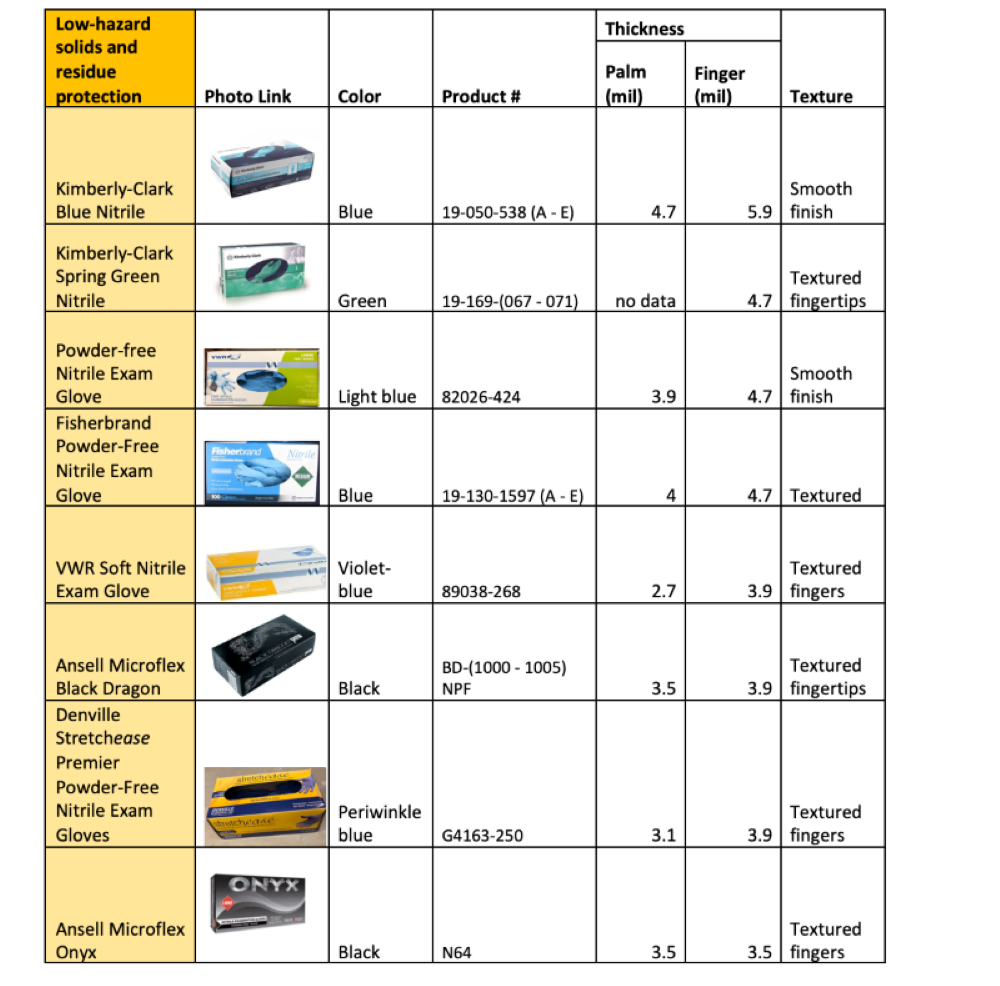
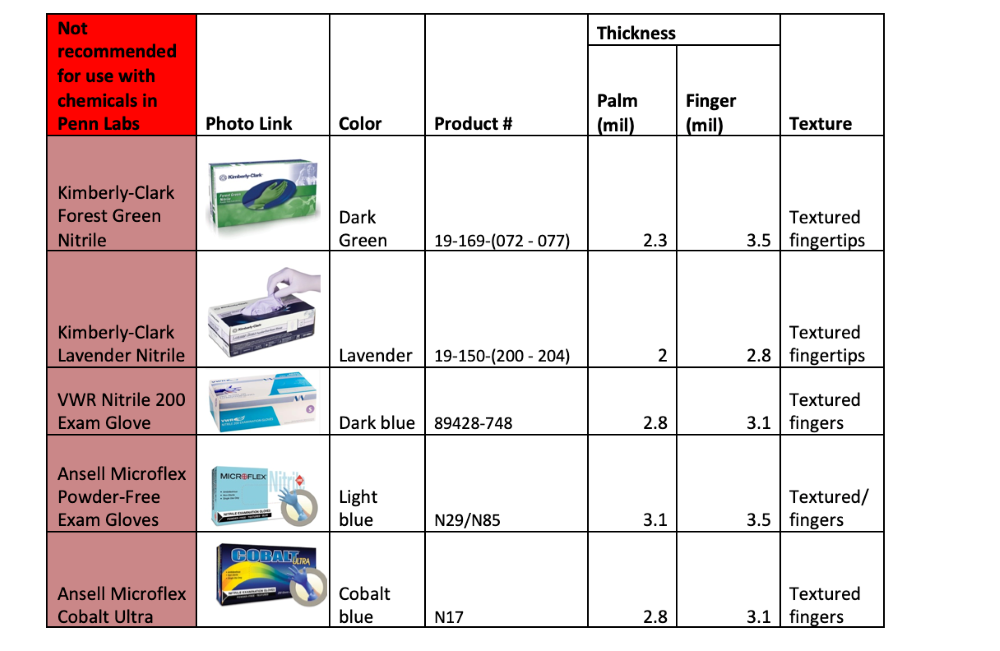
Disposable Nitrile Glove Products
The tables below show some nitrile glove products from common vendors. They are grouped by recommended use type based on the glove thickness and the manufacturer's chemical-compatibility testing data (for those who provided it).
The general limitations and chemical incompatibilities described above apply to all gloves listed below.
This is not an exhaustive list of available gloves. Please consult the manufacturer's product data sheets for complete specifications.
Responsibilities for Research Faculty > Lab Coat, Gloves and Safety Eyewear Policy
Penn's Chemical Hygiene Plan > Section V: Chemical Handling
- Manufacturer websites and product data sheets (various)
- Grainger Quick Tip #212: Glove, Clothing and Material Chemical Compatibility Resources
- Grainger Quick Tip #191: Chemical Protective Gloves
- Phalen, R. N. (2018, December). Not All Gloves are Created Equal. The Synergist, pp. 26 - 29.